Diamonds were first found accidentally and given to children as playthings, but then were recognized as having greater value and were mined in many locations including Brazil and Borneo and India.
Plato believed that the diamond was the kernel of auriferous matter–its purest and noblest pith condensed into a transparent mass. Pliny calls “adamus,” the diamond, a nodosity of gold, and the Rev. C. W. King observes that he may have stumbled on this truth by accident; but it still remains the fact that all diamond mines of which we know anything have been brought to light in the pursuit of gold. This was notably the case in Brazil, and is beginning to be true of the Australian diggings, which Mr. King thinks will yield a vast supply when their gravel comes to be turned over by people having other eyes for other objects than nuggets and gold flakes.
Four thousand years of the world’s history had elapsed before it was ever dreamed that diamonds existed, save in one spot, and that of limited extent. The first diamond of well-ascertained water brought to light out of India was, it is said, accidentally discovered by a miner in Brazil, in the commencement of the eighteenth century. Previous to this, the only known diamonds had been found in Borneo and Hindostan.
Found Accidently
Some Brazilian miners in the beginning of the eighteenth century, while searching for gold, found some curious “pebbles,” which they carried home to their masters as curiosities. Not being considered of any value, they were given to the children to play with. An officer who had spent some years in the East Indies saw these pebbles, and sent a handful to a friend in Lisbon to be examined. They proved to be diamonds, and were pronounced to be equal to those of Golconda.
Strange and various were the vicissitudes attending the early discovery of the Brazilian diamonds. Colossal fortunes were made, and as quickly dissipated. The adventurers who flocked to the diamond grounds saw before them a boundless source of wealth, and they had some reason for their expectations, for, it is said, that in the first twenty years of exploration, Europe received from Brazil diamonds amounting in weight to upwards of three millions of carats–a circumstance that appears almost fabulous, but the mines were then in their rich abundance, and the buried spoil of many ages.
Once diamonds were discovered in Brazil, Portugal declared the diamond district to be crown property and were sent to the great jewel market of Europe, the Netherlands.
Captain Burton, in his “Highlands of the Brazil,” says “that the first man who sent diamonds to Portugal was one Sebastino Leme do Prado, in 1725. He had washed several brilliant octahedrons in the Rio Manso, an influent of the Sequitinhonha. They found no sale; and the same happened to Bernardo (or Bernardino) da Fonseca Lobo, who hit upon a large specimen amongst others, in the Cerro do Frio. There is a local tradition that the latter was a friar who had been in India, and that about 1727, seeing the curious brilliant little stones used as counters at backgammon by the gold miners of the Sequitinhonha, he made a collection of them, and took them to Portugal. Others attribute the discovery to an Ouvidor, or Auditor Judge, fresh from service at Goa. The specimens were sent to the Netherlands, then the great jewel-market of Europe.”
The official account of the diamond exploitation in Brazil was that of D. Lourenco de Almeida, the first governor of Minas Geraes (Aug. 18th, 1721, Sept. 1st, 1732), who reported the new source of wealth to the home government. Portugal at once declared the diamond district to be crown property, and established the celebrated Diamantine demarcation, forty-two leagues in circumference, with a diameter of fourteen to fifteen leagues.
Among the crown jewels of Portugal is a magnificent diamond, “the Braganza,” which was extracted from the mine of Caetha Mirim, in 1741. It was worn by D. Joas VI., who had a passion for precious stones, and possessed them to an amount estimated at three millions sterling. There are some differences as to the weight of this diamond; Mawe and the Abbe Reynal make it 1,680 carats. It is, however, suspected to be a fine white topaz, a stone which, in the Brazil and elsewhere, often counterfeits the diamond. Mr. St. John, in his “Forests of the East,” mentions a noble in Brunei who, for one thousand pounds, offered a diamond for sale the size of a pullet’s egg, which proved to be a pinkish topaz.
These Brazilian discoveries of diamonds are very curious. The Abaete brilliant was found in 1791, and the circumstances of its discovery are related by Mawe and others. Three men, convicted of capital offences, Antonio da Sousa, Jose Felis Gomes, and Thomas da Souza, were exiled to the far west of Minas, and forbidden, under pain of death, to enter a city, wandered about for some six years, braving cannibals and wild beasts, in search of treasure. Whilst washing for gold in the Abaete river, which was then exceptionably dry, they discovered this diamond, weighing nearly an ounce (576 grains–144 carats). They trusted to a priest, who, despite the severe laws against diamond-washers, led them to Villa Rica, and submitted the stone to the Governor of Minas, whose doubts were dissipated by a special commission. The priest obtained several privileges, and the malefactors their pardon, no other reward being mentioned.
Diamonds are minerals. They are one of two crystalline forms of the element carbon the hardest natural substance known, used as a gem and in industry.
Diamonds crystallize in the isometric system commonly as transparent to translucent white, colorless, yellow, green, blue, or brown octahedrons (the familiar diamond shape). The extraordinary brilliancy of diamonds after faceting is due to their very high refractive index, which is greater than that of any other naturally occurring gemstone. In addition to the gem varieties there are bort, which is poorly crystallized or of inferior color and in fragmentary condition, and carbonado (black diamond), which is gray to black and opaque, with poor cleavage. Bort and carbonado are used as abrasives, in the cutting of diamonds, and for the cutting heads of rock drills.
Diamonds are found in alluvial (loose earthy material deposited by running water) formations and in volcanic pipes, filled for most of their length with blue ground or kimberlite, an igneous rock consisting largely of serpentine. At the surface the blue ground is weathered to clay called yellow ground. Diamantiferous (or diamondiferous), or diamond-yielding, earth is mined both by the open-pit method and by underground mining. After being removed to the surface, it is crushed and then concentrated. Sorting is done by passing the concentrated material in a stream of water over greased tables. The diamond, being largely water repellent, sticks to the grease, but the other minerals retain a film of water, which prevents them from adhering to the grease. The diamonds are then removed from the grease, cleaned, and graded for sale.
Synthetic diamonds were successfully produced in 1955; a number of small crystals were manufactured when pure graphite mixed with a catalyst was subjected to pressure of about 1 million lb per sq in. and temperature of the order of 5,000 (3,000). Synthetic diamonds are now extensively used in industry.
The discoveries of 1870-71 in South Africa led to a great number of prospectors staking out claims and securing the diamonds by open-pit or quarry mining. The damage caused by floods and mudslides, unavoidable when there were so many different claims, was an important factor in the series of amalgamations carried on by Cecil Rhodes and Barnett Barnato. Rhodes brought about the merging of their interests in the De Beers Consolidated Mines, Ltd., which established (1889) an effective monopoly over the diamond industry. Loss of diamonds by theft was reduced through the passage of the so-called I.D.B. (Illicit Diamond Buying) Act, which limited the trade to licensed buyers and imposed penalties for the possession of uncut stones without a license. Thefts were further curtailed by the institution of compounds in which the workers live while employed by the company and which they leave only after being thoroughly searched.
Diamonds are said to be a woman’s best friend. Diamonds are forever!
Diamond is the allotrope of carbon where the carbon atoms are arranged in an isometric-hexoctahedral crystal lattice. Its hardness and high dispersion of light make it useful for industrial applications and jewelry. It is the hardest known naturally-occurring mineral. It is possible to treat regular diamonds under a combination of high pressure and high temperature to produce diamonds (known as Type-II diamonds) that are harder than the diamonds used in hardness gauges.Presently, only aggregated diamond nanorods, a material created using ultrahard fullerite (C60) is confirmed to be harder, although other substances such as cubic boron nitride, rhenium diboride and ultrahard fullerite itself are comparable.
Diamonds are specifically renowned as a material with superlative physical qualities; they make excellent abrasives because they can be scratched only by other diamonds, borazon, ultrahard fullerite, rhenium diboride, or aggregated diamond nanorods, which also means they hold a polish extremely well and retain their lustre. Approximately 130 million carats (26,000 kg (57,000 lb)) are mined annually, with a total value of nearly USD $9 billion, and about 100,000 kg (220,000 lb) are synthesized annually.
The name diamond derives from the ancient Greek ἀδάμας (adamas) “invincible”, “untamed”, from ἀ- (a-), “un-” + δαμάω (damáō), “to overpower, to tame”. They have been treasured as gemstones since their use as religious icons in ancient India and usage in engraving tools also dates to early human history. Popularity of diamonds has risen since the 19th century because of increased supply, improved cutting and polishing techniques, growth in the world economy, and innovative and successful advertising campaigns. They are commonly judged by the “four Cs”: carat, clarity, color, and cut.
Roughly 49% of diamonds originate from central and southern Africa, although significant sources of the mineral have been discovered in Canada, India, Russia, Brazil, and Australia. They are mined from kimberlite and lamproite volcanic pipes, which can bring diamond crystals, originating from deep within the Earth where high pressures and temperatures enable them to form, to the surface. The mining and distribution of natural diamonds are subjects of frequent controversy such as with concerns over the sale of conflict diamonds (aka blood diamonds) by African paramilitary groups.
In India
Although Golconda is associated with famous diamond beds, the diamonds found at Parteal were merely cut and polished at Golconda and the place itself is now in ruins.
So far, the new world gave promise of a more copious supply of diamonds than the old. The famous jewels of Golconda are associated with the diamond beds of Raolconda and Gomec Parteal, from whence they were really derived, and which are situated in the territories of the kings of Golconda, on the north bank of the Kistna river. The fickle changes of fortune, which are especially obvious in the discoveries of the precious metals and jewels, are conspicuous in this world-renowned district, which has been the fountain of almost fabulous wealth. The diamonds found at Parteal were merely cut and polished at Golconda; the place itself now affords no indication of its former distinction, being in ruins, and the inhabitants, descendants of those who were enriched by their precious discoveries, being ill-clothed and half-starved in appearance.
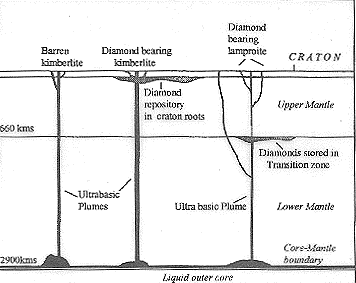
Diamond, known in India since the Vedic times (2000 BC), is pure carbon formed at high pressure that exists in the earth’s mantle zone. When subjected to pressure >5 GPa (>50,000 atmospheres) and temperature of 1600 K (2400°F), prevailing at depths of 200 km, the carbon is transformed to diamond, and it remains stable there for millions of years. A number of processes are envisaged to operate for diamond nucleation from methane, carbon dioxide or particulate carbon under favourable pressure and temperature conditions aided by catalysts (COS, CS2, N2)1. From the diamond stability zone in the mantle, they are brought later to the surface by plumes of mantle melt material that ascend with explosive rapidity (Figure 1). This rapid ascent prevents diamond to return to graphite structure or its oxidation. Weathering of the host rocks liberates the diamonds, which soon get dispersed by streams and rivers to form alluvial deposits. Diamonds of non-mantle origin (crustal diamonds) are observed within garnets and zircons in calc-silicate rocks, amphibolites and pyroxenites. Here, diamonds are believed to have formed in situ at high pressure from carbon derived from the recycled (subducted) crust2,3. Microscopic diamonds are seen in some meteorites and their impact craters as well as within larger diamondiferous pipes along with macrodiamonds. Some consider that these crystals had little time to grow to macro-size, before transportation to the surface or they may have evolved under sparse carbon availability leading to stunted growth or due to resorption of larger diamonds.
links:Wikipedia
Diamonds in komatiites and some contemporary views about this gem-carbon